If you received a Cartiva synthetic cartilage implant in your big toe and experienced persistent pain, implant failure, or required a second "revision" surgery, you are not alone. Thousands of patients are now learning that the device marketed as a better alternative to toe fusion may have been defectively designed.
As of February 2026, a federal multidistrict litigation (MDL) has been established to consolidate these mass-tort claims. Before you decide on your next steps, here are the five key takeaways regarding the Cartiva implant lawsuit:
- A Federal MDL is Active: Claims are now centralized in MDL No. 3172, allowing for streamlined discovery against the manufacturer.
- The Device Was Recalled: Stryker issued a Class 2 Device Recall for all Cartiva SCI sizes due to higher-than-expected failure rates.
- High Failure Rates: Post-market data suggest the implant may sink into the bone (subsidence) or fracture, often requiring the very fusion surgery it was meant to avoid.
- Failure to Warn: Lawsuits allege the manufacturer knew about these risks but failed to disclose them to the FDA, surgeons, or patients.
- Strict Filing Deadlines: Statutes of limitations in Alabama and Tennessee mean that waiting too long to consult a mass tort lawyer could permanently bar your right to recovery.
Understanding Cartiva Toe Implants
What is a Cartiva Synthetic Cartilage Implant?
The Cartiva Synthetic Cartilage Implant (SCI) is a small, cylindrical hydrogel polymer device. It was granted FDA premarket approval in 2016 to treat hallux rigidus, a painful form of degenerative arthritis at the base of the big toe. The device was intended to act as a synthetic substitute for damaged cartilage, theoretically preserving the joint's range of motion.
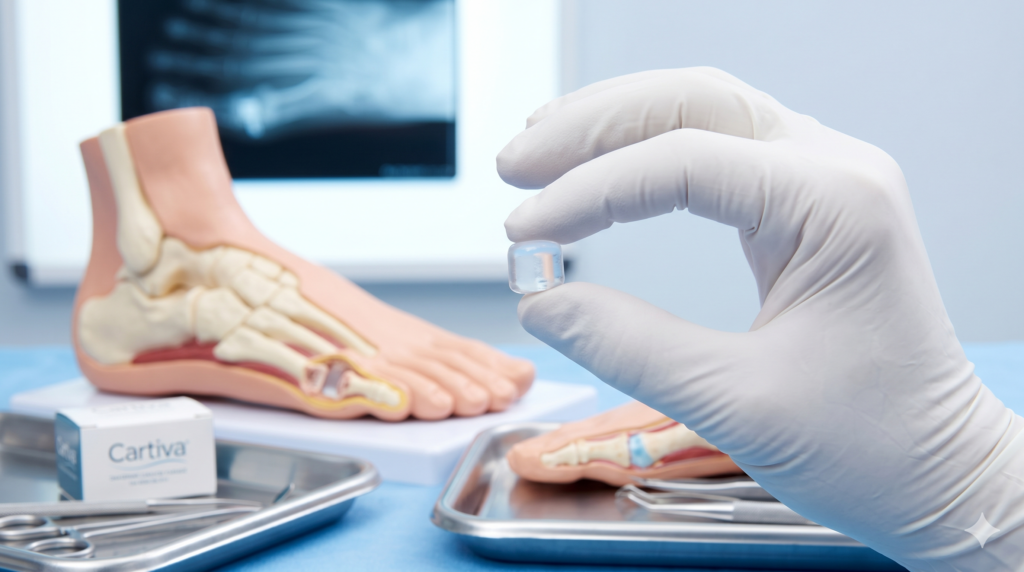
How Does the Cartiva Implant Work?
The implant is "press-fit" into the head of the first metatarsal bone. Unlike other orthopedic implants, it does not use screws or adhesives. It was marketed as a middle ground between conservative treatment and "fusion" surgery (arthrodesis), which permanently joins the bones together and eliminates movement.
The Cartiva Implant Recall
Reasons Behind the Recall
In late 2024, the manufacturer issued an Urgent Medical Device Recall for all lots of the Cartiva SCI. The recall was prompted by post-market reports indicating that the device was failing at rates far higher than those represented during the initial FDA approval process.
Implications for Injured Patients
The recall serves as a formal acknowledgment that the device carried risks of fragmentation, subsidence (sinking into the bone), and displacement. For patients who have already suffered these complications, the recall and the subsequent MDL Transfer Order (MDL No. 3172) provide the legal foundation needed to pursue compensation for medical bills and pain and suffering.
Cartiva Implant Complications
Common Cartiva Implant Failure Symptoms
Patients with a failing Cartiva implant often experience a specific cluster of symptoms:
- Subsidence: The implant sinks into the metatarsal bone, causing bone-on-bone contact.
- Migration: The device moves out of its drilled cavity.
- Persistent Pain: Severe discomfort that often exceeds the pain felt prior to the initial surgery.
- Fragmentation: The hydrogel material breaks apart within the joint.
- Revision Surgery: The necessity of a second procedure to remove the device and perform a toe fusion.
Legal Rights for Plaintiffs
Personal Injury Lawsuits Against Manufacturers
Victims of defective medical devices have the right to file suit based on Strict Product Liability and Negligence. These claims argue that the Cartiva implant was "unreasonably dangerous" as designed and that the manufacturer failed to provide adequate warnings to the medical community.
The Role of Product Liability Attorneys
Mass tort litigation is complex, requiring the analysis of thousands of pages of internal corporate documents and FDA filings.
Navigating a Cartiva Implant Lawsuit
Steps to Take for Defective Device Victims
- Request Your Surgical Logs: Ensure you have the specific lot number and implant size used.
- Monitor Symptoms: Document any loss of mobility or return of pain.
- Consult a Specialist: If failure is suspected, seek an orthopedic opinion regarding revision options.
- Contact Legal Counsel: Ensure your claim is filed within the statute of limitations.
Potential Cartiva Implant Lawsuit Settlement Amounts
While no outcome is guaranteed, settlements in medical device mass torts typically account for the costs of revision surgeries, lost wages, and permanent physical impairment. The formation of MDL No. 3172 is a significant step toward a potential global resolution for injured plaintiffs.
About Our Firm:
Cory Watson Attorneys is a Birmingham, Alabama-based firm with offices in Nashville and Memphis, TN. We have fought for the rights of the injured for over 40 years, specializing in complex mass-tort and product-liability litigation.
Disclaimer: This article is for informational purposes only and does not constitute legal advice. Past results do not guarantee future outcomes.